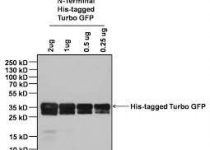
Cusabio Influenza A virus Recombinant
What is the recombinant flu vaccine?
Recombinant influenza (flu) A virus vaccine are produced using recombinant technology. This method does not require an egg-grown vaccine virus and does not use chicken eggs in the production process. The recombinant influenza vaccine was first licensed by the FDA in the United States in 2013. Currently, recombinant influenza vaccine and cell culture-based influenza vaccine are the only egg-free influenza vaccines licensed for its use in the United States. A quadrivalent recombinant influenza vaccine (Flublok Quadrivalent) is available for the 2021-2022 influenza season.
Who can receive Flublok Quadrivalent?
Flublok Quadrivalent is licensed for use in persons over 18 years of age.
Who should not receive Flublok Quadrivalent?
People who are not within the FDA-approved age range (under 18 years of age) and who have had severe allergic reactions to any ingredient in the vaccine should not receive Flublok Quadrivalent.

What are the possible benefits of recombinant influenza vaccines?
Because the recombinant influenza vaccine is not dependent on the supply of eggs, the manufacturing process for the recombinant influenza vaccine could be faster than that of egg-based vaccines in the event of a pandemic or egg shortage needed to grow influenza viruses. This process is not limited by the selection of vaccine viruses that are adapted to grow in eggs. It also prevents mutations that can occur when viruses are grown in eggs, which can sometimes affect how well the finished vaccine works.
A 2019 study in Hong Kong showed that vaccination against recombinant influenza resulted in greater antibody response in adults aged 65 years and older compared to traditional influenza vaccines. Although a higher antibody response after influenza vaccination does not guarantee better clinical protection, further studies are warranted to determine the potential benefits of recombinant influenza vaccines in this age group.
Purity: greater than 90% as determined by SDS-PAGE.
Target names: NP
Uniprot No.: P16980
Research Area: Others
Alternative Names: NP Nucleoprotein; nucleocapsid protein; Protein N
Species: Influenza A virus (strain A/Equine/Prague/1/1956 H7N7)

Source: Yeast
Expression Region: 1-498aa
Mole Weight: 58.2 kDa
Protein Length: Total length
Tag Information: N-terminal 6xHis-tagged
Form: Liquid or Lyophilized Powder
Note: We will preferably ship the format we have in stock, however, if you have any special requirements for the format, please remark your requirement when placing the order, we will prepare according to your demand.
Buffer
If the dosage form is liquid, the default storage buffer is Tris/PBS based buffer, 5%-50% glycerol.
Note: If you have any special requirements for glycerol content, please remark it when you order. If the administration form is a lyophilized powder, the buffer before lyophilization is Tris/PBS-based buffer, 6% trehalose, pH 8.0.
Reconstitution
We recommend that this vial be briefly centrifuged before opening to bring the contents to the bottom. Reconstitute protein in sterile deionized water at a concentration of 0.1-1.0 mg/mL. We recommend adding 5-50% glycerol (final concentration) and an aliquot for long-term storage at -20°C/-80°C. Our final default glycerol concentration is 50%. Customers could use it for reference.
Storage Conditions
Store at -20°C/-80°C upon receipt, need to be aliquoted for multiple uses. Avoid repeated cycles of freezing and thawing.
Shelf life
Shelf life is related to many factors, storage condition, buffer ingredients, storage temperature and the stability of the protein itself. Generally, the shelf life of the liquid form is 6 months at -20°C/-80°C. The shelf life of the lyophilized form is 12 months at -20°C/-80°C.
Delivery time
The delivery time may differ depending on the form or location of purchase, consult your local distributors for the specific delivery time.
Notes: Repeated freezing and thawing is not recommended. Store working aliquots at 4°C for up to one week.